Objective:
The objective of this project was to create a hand warmer using water and one type of salt. The main goals of the hand warmer were:
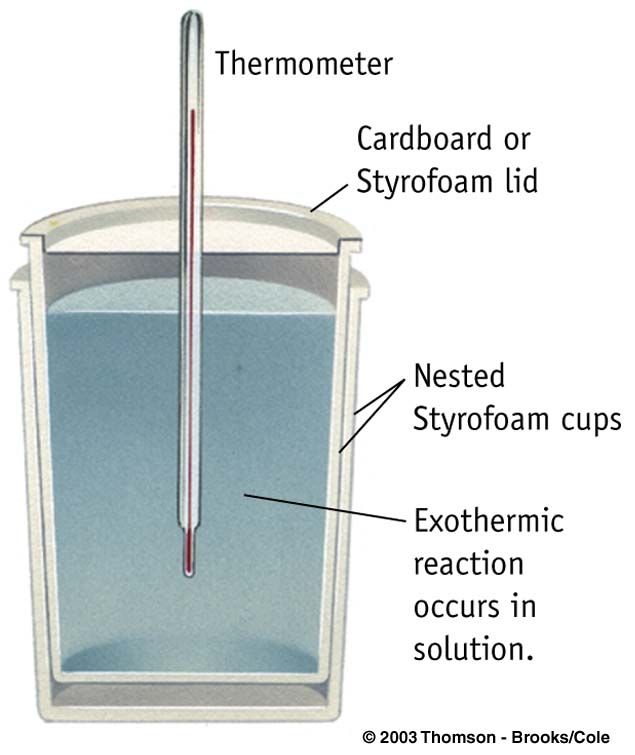
We started off by testing three different salts to determine which one had the highest temperature change. To determine this, we used a coffee cup calorimeter (image below). The coffee cup calorimeter works by placing the chosen salt and water into a styrofoam cup and them placing styrofoam over the top of the cup. From there, a thermometer is placed into the cup to determine temperature change.
The objective of this project was to create a hand warmer using water and one type of salt. The main goals of the hand warmer were:
- Temperature increase of 20*C
- Have a volume around 50 mL
- Costs as little as possible to make
- Uses chemicals that are safe
- Is as environmentally friendly as possible
We started off by testing three different salts to determine which one had the highest temperature change. To determine this, we used a coffee cup calorimeter (image below). The coffee cup calorimeter works by placing the chosen salt and water into a styrofoam cup and them placing styrofoam over the top of the cup. From there, a thermometer is placed into the cup to determine temperature change.
Once we determined which salt had the highest temperature change, for us that salt was CaCl2, we moved onto calculations to determine the calorimeter constant and how much CaCl2 we should use to have the best results. A picture of our calculations is pictured below.
Why we chose CaCl2
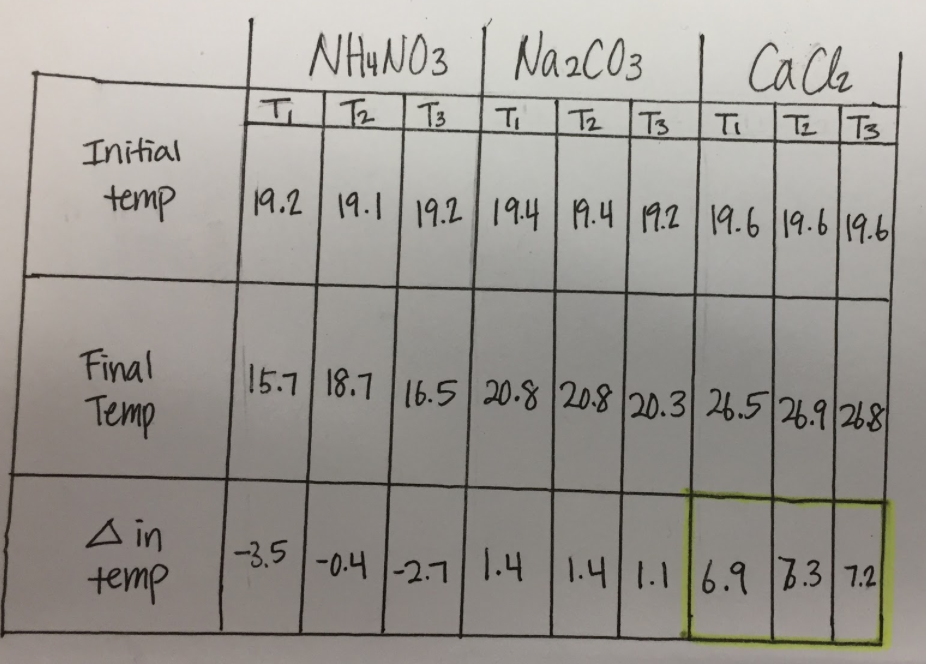
We decided to use CaCl2 because it had the highest temperature change. Our data table, pictured below, shows all trials for all three salts we tested (NH4NO3, Na2CO3, and CaCl2)
We decided to use CaCl2 because it had the highest temperature change. Our data table, pictured below, shows all trials for all three salts we tested (NH4NO3, Na2CO3, and CaCl2)
Our presentation for the hand warmer challenge is pictured below.
Our Hand Warmer
We used 30 mL of water and 7 g of CaCl2. We also used aluminum foil to maintain the heat for a longer time. When tested, it took a full 45 minutes for the hand warmer to return to its original temperature after it was activated. Parts of our hand warmer are pictured below.
We used 30 mL of water and 7 g of CaCl2. We also used aluminum foil to maintain the heat for a longer time. When tested, it took a full 45 minutes for the hand warmer to return to its original temperature after it was activated. Parts of our hand warmer are pictured below.
The Chemistry:
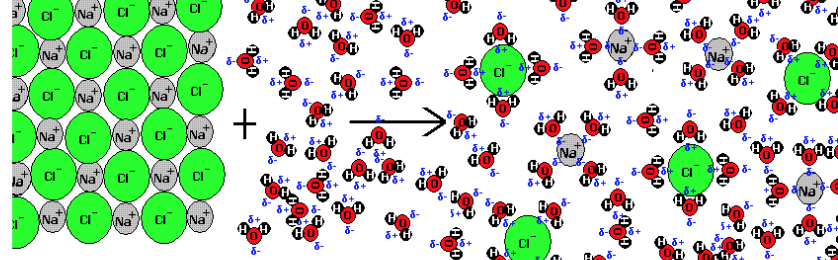
- Breaking bonds absorb energy from its surroundings, while forming new bonds and releasing energy to the surroundings (exergonic reaction).
- When an ionic solid dissolves in water, ionic bonds between cations and anions and hydrogen bonds between water molecules break, and new attractions are formed.
Reflection:
This project was pretty straightforward. While still in the testing phase, we didn't run into any significant problems. But once we got to the calculations, that all changed. Our calculations repeatedly came out way out of the range that was expected. To stop this problem, we had to significantly change out calorimeter constant to receive a more logical answer. Another problem we ran into was our design. Our design was quite basic, a bag inside another bad, but we ran into problems of leakage. To solve this problem, we made sure to seal our bags perfectly to prevent accidental leaking. We also added multiple layers to our design to make sure that even if the bag did leak, it wouldn't spill onto the consumer's hand. Two things I liked were the experimentation and learning how to create an effective hand warmer. Two things I found frustrating was the constant trial and error and the consistent leaking of our design. Some things I improved upon were my chemistry knowledge and design building skills. Something we could improve upon was making sure our data was accurate the first time so we wouldn't have to redo calculations. Another thing we could've improved upon was our presentation, as we could've added more chemistry.